1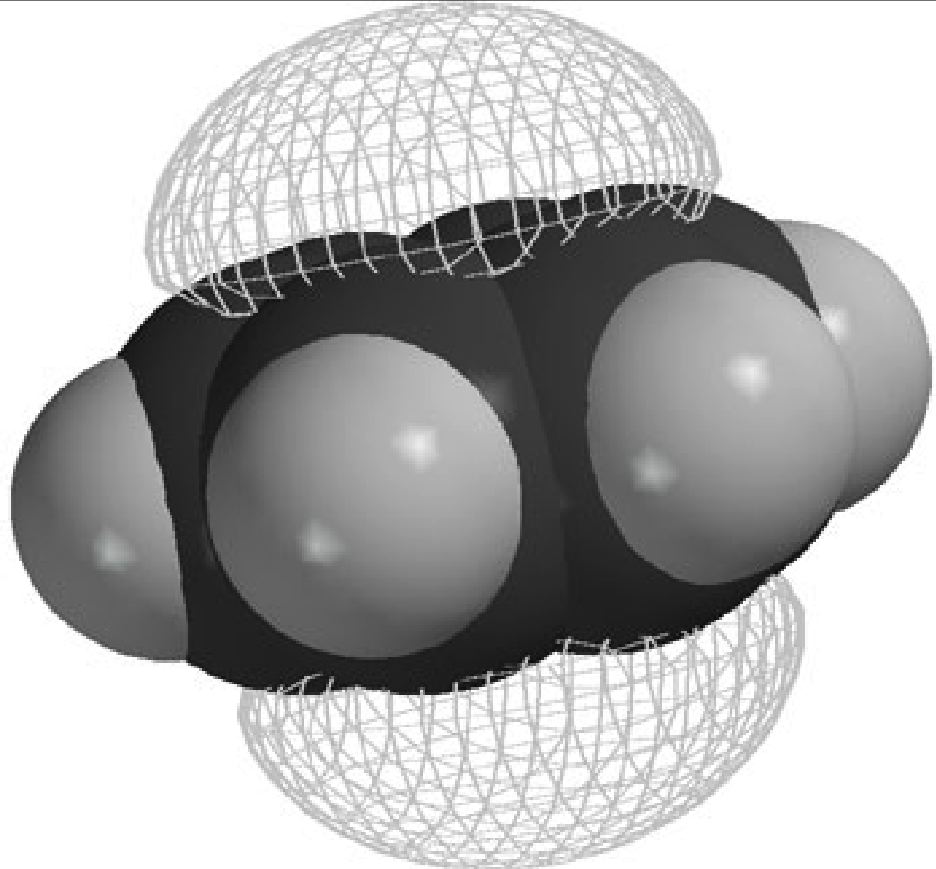
2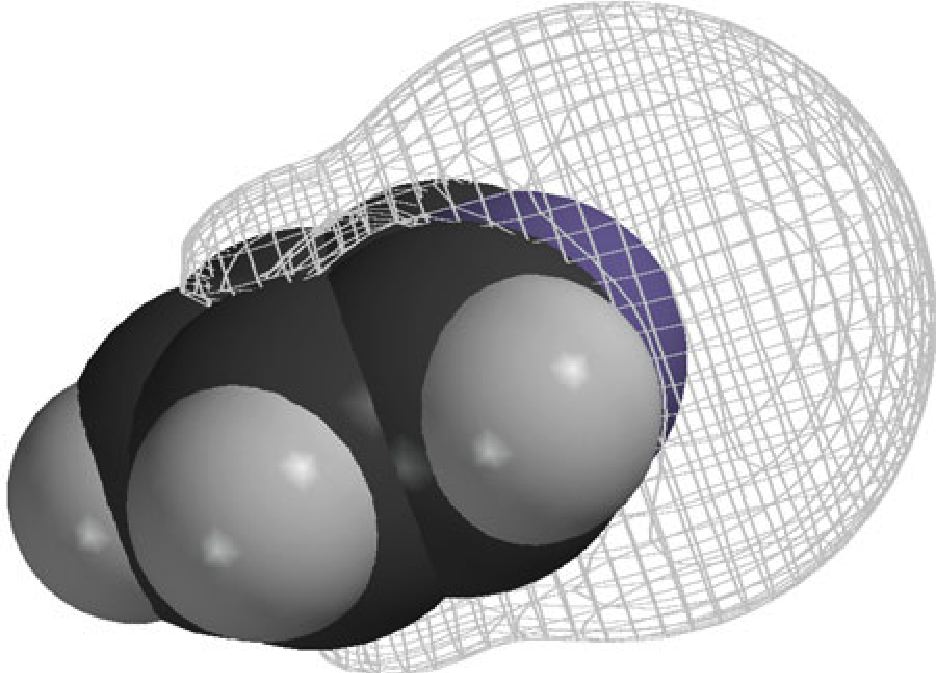
E-mail ert@sas.upenn.edu
We have found substantial differences in electrostatic potential surfaces (shown in 1 and 2) between different aromatic systems such as benzene (1) vs. pyridine (2). These differences correlate nicely with binding properties of glucose-based peptidomimetics containing different aromatic substituents, and they appear to explain observed differences in binding to the somatostatin receptor.
1
2
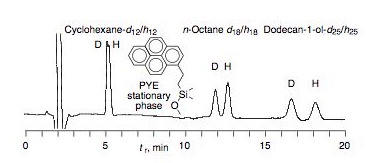
We have studied the separation of hydrogen/deuterium isotopologue pairs by means of reversed-phase chromatographic separation in order to examine deuterium isotope effects on hydrophobic binding. The results (see Figure, where tr is the HPLC retention time) demonstrate that dispersion interactions in the hydrophobic phase are an important component of hydrophobic interactions.
We are also interested in design of extended three-dimensional structures with specific architectures and novel properties. Large molecules with architecturally complex structures and shapes, having properties designed for specific purposes, are of increasing importance in all areas of organic and bioorganic chemistry.
A major area of interest in our lab has been study of interactions, transition structures, and mechanisms, for example, mechanistic studies on deuterium and sulfur isotope effects, solvolysis reactions, E2 eliminations, mass spectra, electrical discharge reactions, acid-base catalysis, the Diels-Alder reaction, theory of transition state structural effects, and reactions of carbenes.
Previous bioorganic studies in our lab involved structures and interactions of saccharides and glycolipids utilizing 13C NMR relaxation times, hydrophobic interactions, carbene photochemical labeling of model membranes, and proteins of synaptic vesicles.
Back to ET Homepage
Penn Chem Library
Back to Chemistry Homepage