Unit for Experimental Psychiatry, Institute of Pennsylvania Hospital and University of Pennsylvania Medical School, 111 North 49th Street, Philadelphia, Pennsylvania, 19139 U.S.A.
INTRODUCTION
Over 10 years ago we proposed the use of napping as a countermeasure to performance and mood/ degradation due to work scenarios that extend beyond the usual 8-hr. shift, or that involve sleep loss, or that require performance quasicontinuously over many days. Since our early reports on napping behavior (Evans et al. 1977; Dinges et al. 1980), studies of napping have been conducted in non-sleep-deprived subjects (Taub 1977); in shiftworkers (Akerstedt and Torsvall 1985.); in time-free environments (Zulley and Campbell 1985); and in sleep loss protocols with performance demands (Naitoh 1981; Haslam 1981; Gillberg 1984; Webb 1985).
Studies of napping during sleep loss usually show that it can improve performance and mood relative to no sleep at all. It is not clear, however, whether this effect varies with: (1) the degree of sleep loss prior to the nap; (2) the time-of-day (circadian phase) of the nap; (3) the length and sleep stage composition of the nap; (4) the type, timing, and duration of the performance; or (5) individual differences in sleep behavior. This paper describes a study that addresses some of the factors relevant to the benefits of naps during sustained periods of human functioning.
MATERIAL AND METHODS
Five groups of healthy, young adults (8 per group) were permitted 2-hr. nap opportunities placed near either the
23
24
circadian peak (P naps; circa 1500 hrs.) or trough (T naps; circa 0300 hrs.) in body temperature and preceded by either 6(P), 18(T), 30(P), 42(T), or 54(P) hrs. of prior wakefulness. The naps took place during a 54-hr. period of quasicontinuous work. Every 2.5 hrs. throughout this 54-hr. period subjects worked on cognitive and motor tasks in performance bouts lasting 45 mins.; in between bouts they were permitted to read, play games, and engage in conversation, but not sleep. The environment was free of all time cues (windows occluded, no clocks, radios, or televisions). Subjects were run in same-sex pairs, they were monitored continuously throughout the 54-hr. trial, they did not know when the naps would occur or how long they would last. Nap sleep was recorded polygraphically and sleep stage composition was scored. The effects of the naps on a simple, 10-minute, visual reaction time (VRT) task, self-reported sleepiness, and oral temperature were evaluated by comparing the temporal pattern of these variables during the 54-hr. period among the five groups. A special analytic approach was developed to evaluate VRT performance (Dinges and Powell in press); it included the mean of reciprocal VRT data from the slowest 10% of responses within a trial, taken as a percentage of baseline performance.
RESULTS
Sleep stage composition of the naps changed as a function of prior sleep loss. For most variables this change was curvilinear; greatest across the first day of sleep loss (P-6 to P-30) and reaching asymptotic levels by the T-42 nap. Subjects took longer to go to sleep, slept less, and had less SWS during the P-6 nap relative to all other nap conditions.
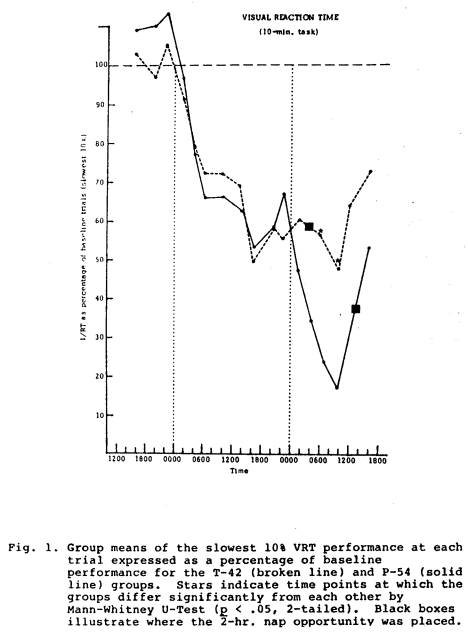
VRT performance was compared between the P-54 control group and each of the other four groups (Figures 1-4). The comparison between T-42 and P-54 groups revealed identical performance functions until after the T-42 group napped, at which time this group averaged significantly better
25
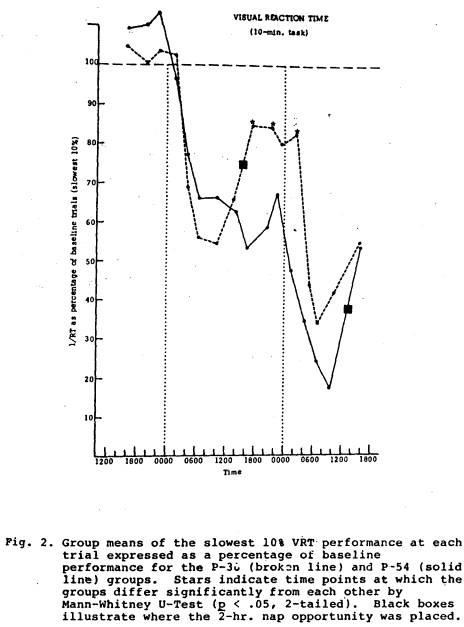
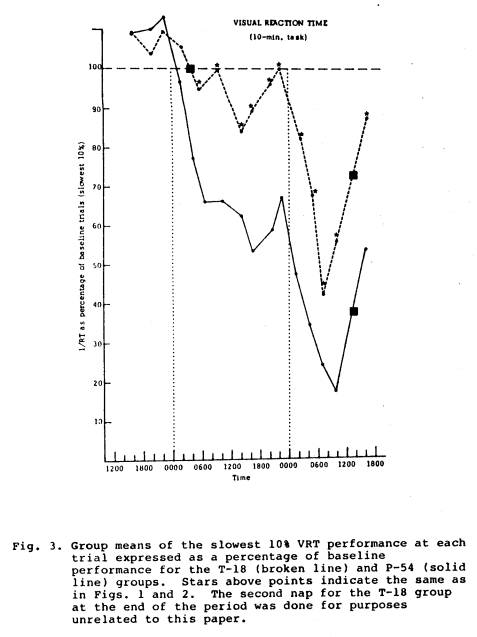
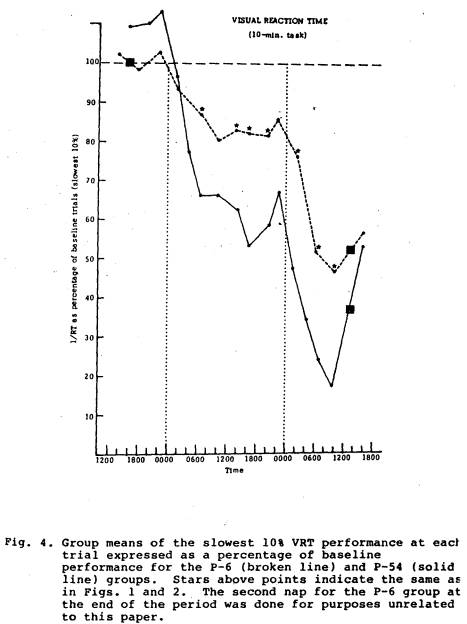
performance for the next 10 hrs. than did the P-54 group (Fig. 1). The comparison between the P-30 group and the P-54 group also showed similar functions until after the P-30 nap on Day 2, when that group did significantly better for about 10 hrs than did the P-54 group (Fig. 2). Compared to the P-54 group the T-18 group function was not only higher after the nap, but it remained significantly higher for over 24 hours (Fig. 3). The P-6 data compared to the P-54 pattern revealed difference between the two groups, but not until approximately 12 hours after the P-6 nap; for about 24 hours after this the P-6 group did significantly better than the P-54 group (Fig. 4).
When similar comparisons were made between groups for sleepiness ratings and oral temperature readings, no differences were found. The naps did not significantly alter the increasing sleepiness or decreasing temperature readings during the 54-hr. scenario, although like performance, both classes of data showed a circadian modulation.
CONCLUSIONS
In summary this study shows that a 2-hr. nap can prevent some of the performance deterioration seen in subjects otherwise deprived of sleep opportunities. Curiously, this gain is not manifest in subjects' mood reports. In terms of the variables that might moderate the effects of a nap, these data suggest that neither circadian phase of the nap (P vs T), nor the length of prior sleep loss, nor the sleep stage composition of the nap, relate to the performance benefits derived from the nap. Rather, obtaining sleep at any time seems to help performance. This is the first study that has fully assessed each of these variables. As predicted by Prof. Orne, the beneficial effect of a nap on VRT performance occurred even when the nap was taken before any sleep debt accumulation, and despite the fact that subjects were not aware of any benefit. When viewed this way, naps can serve a "prophylactic" function as well as a "recovery" function.
26

27
28
29
30
REFERENCES
Akerstedt T, Torsvall L (1985) Napping in shiftwork. Sleep 8: 105-109
Dinges DF, Orne MT, Orne EC, Evans FJ (1980) Voluntary selfcontrol of sleep to facilitate quasi-continuous performance. U. S. Army Medical Research and Development Command, Maryland (NTIS No. AD-A102264) 222 pp
Dinges DF, Powell JW (in press) Microcomputer analyses of performance on a portable, simple visual RT task during sustained operations. Beh Res Meth Instr Comp
Evans F, Cook M, Cohen H, Orne EC, Orne MT (1977) Appetitive and replacement nap: EEG and behavior. Science 197:687-689
Gillberg M (1984) The effects of two alternative timings of a 1-hour nap on early morning performance. Bio Psychol 19:45-54
Haslam D (1981) The military performance of soldiers in continuous operations: Exercise Early Call I and II. In:Johnson L, Tepas D, Colquhoun W, Colligan M (EDs) Advances in Sleep Research, vol 7. Spectrum, New York pp 435-458
Naitoh P (1981) Circadian cycles and restorative power of naps. In:Johnson L, Tepas D, Colquhoun W, Colligan M (EDs) Advances in Sleep Research, vol 7. Spectrum, New York pp 553-580
Taub JM (1977) Napping behavior, activation and sleep function. Waking and Sleeping 1:281-290
Webb WB (1985) Experiments on extended performance: Repetition, age, and limited sleep periods. Beh Res Meth Instr Comp 17:27-36
Zulley J, Campbell S (1985) Napping behavior during "spontaneous internal desynchronization": sleep remains in synchrony with body temperature. Human Neurobio 4:
ACKNOWLEDGEMENTS
The present study was supported by U. S. Office of Naval Research contract N00014-80-C-0380 to DFD, and in part by a grant from the Institute for Experimental Psychiatry. We thank DI Tepas, SR Fairbrother, C Auxier, JW Powell, AM Whitehouse, MF Auxier, CK Dinges, J Moore, and R Barras.