|
The work of Maxwell Planck highlighted the concept that
light had properties of both waves and particles. The energy of
the light particle (photon) is quantized, each particle of light
"carrying" with it a quanta of energy. Scientific discoveries
immediately preceding the original publication of C.V. Raman's
historical experiment demonstrated that electromagnetic radiation could
be inelastically scattered as a result of interaction with
molecules. An inelastic scatter results when some of the energy
of the electromagnetic radiation put into a molecule is absorbed, the
remaining portion of the energy is emited. The
resulting energy emission from the molecule is lower in energy than the
original. In 1928 C.V. Raman, in collaboration with K.S.
Krishnan, published their light scattering experimental results in Nature. It was in this
article that Raman explained the basis for the Raman Spectroscopy
theory: molecular transitions caused by incident light will
inelastically scatter the light. (16) To read
the text of Sir Raman's original article, click on this link. |
 Photo 7: C.V. Raman http://www.deltanu.com/tutorial.htm |
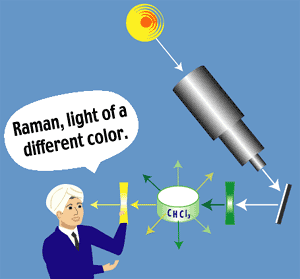 Figure 14: A light of a different color http://www.deltanu.com/tutorial.htm |
In his original experiment, Raman focused a beam of sunlight and passed
this beam through a green filter. The green light was then passed
through a solution of chloroform. Raman was able to detect a
faint yellow emission of light from the chloroform. The green
light (higher energy) was inelastically scattered by the chloroform and
the resulting emission of yellow light (lower energy) was his
proof. As a result of this ground-breaking work, Raman received
the Nobel Prize. Raman Spectroscopy and Spectrophotometers have come a long way since Sir Raman performed his original experiments. This analytical technique was facilitated by replacing the incident light source (sunlight) with LASER light. Since the intensity of emitted light is a factor of 104 less than the incident light, using a LASER for input greatly enhanced the output signal of the scattered light. (16) |
| Project
Home |
Raman
Spectroscopy |
Searching
For Signs of Life |
The
Raman Spectrophotometer |
Why Mars?
|
References |